Use Instructions
Table Of Contents
- Verasense instructions for use
- description
- INDICATIONS
- ContraINDICATIONS
- WARNINGS
- PRECAUTIONS
- INstructIONS
- VERASENSE troubleshooting
- DECONTAMINATION OF Product returned for complaint investigation
- VERASENSE Device Specifications
- FoR REference only
- Material: White, 24# Paper, 97 Brightness
- Measurements: 8.5” x 11”
- Specs: 4/4 CMYK
- Double sided print; letters out
- Trim, staple upper left corner and fold to 5.5” x 8.5”
- Process Colors:
OrthoSensor, Inc.
1855 Griffin Road
Suite A-310
Dania Beach, FL 33004-2200 USA
www.orthosensor.com
LB-5190 Rev. 6 (EN) 12
08-2020
For further information, contact the OrthoSensor Customer Service Center by phone at + 1 954-577-7770 or by e-mail
at customerservice@orthosensor.com or go to www.orthosensor.com.
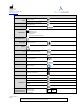
Table 4 - Guidance and manufacturer’s declaration – electromagnetic emissions
The VERASENSE and accessories have been tested and found to comply with the electromagnetic compatibility (EMC) limits for medical
devices IEC 60601-1-2:2014.
Emissions/Immunity Test
Compliance Level
RF Emissions
CISPR 11
Class A, Group 1
Electrostatic discharge (ESD)
IEC 61000-4-2
± 8 kV contact discharge
± 2, 4, 8 & 15kV air discharge
Radiated RF Immunity
IEC 61000-4-3
3 V/m
80 MHz to 2.7 GHz
80% at 1 kHz
Immunity to Proximity Fields from RF wireless communication
equipment
IEC 61000-4-3
28 V/m
Conducted Immunity
IEC 61000-4-6
0.15 - 80 MHz
3 Vrms & 6Vrms in ISM Band 1 kHz, AC Mains
Electrical fast transient/burst
IEC 61000-4-4
± 2 kV AC Mains
5kHz & 100 kHz
Magnetic Immunity
IEC 61000-4-8
30 A/m
Voltage dips, short interruptions and voltage variations on power
supply input lines
IEC 61000-4-11
0% U
T
.5 cycle
0% U
T
1 cycle
70% U
T
25 cycles
0% U
T
5 Sec